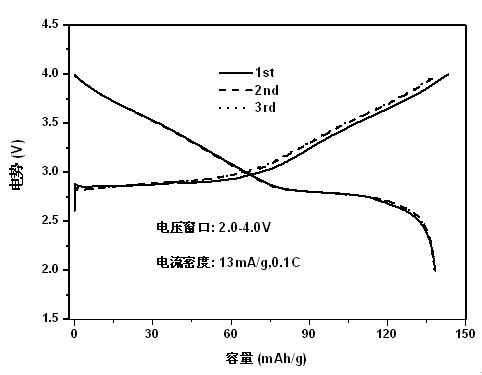
With the large-scale application of portable electronic products, the total amount of lithium metal in the world cannot meet the demand. As promising alternatives to lithium-ion batteries, room temperature sodium-ion batteries are attracting more and more attention for large-scale energy storage applications due to its natural abundance, similar chemical properties and low cost of sodium resources. In our work, in order to get higher capacity titanium is chose to replace manganese to produce a new sodium-ion battery cathode material NaNi0.5Mn0.3Ti0.2O2 which is synthesized by the solid state reaction In our research this material exhibit excellent performance with an initial discharge capacity of 138 mAh g-1 and coulombic efficiency of above 96% in sodium-ion half-cell. The performance of sodium-ion full-cells with NaNi0.5Mn0.3Ti0.2O2 as the cathode and presodiated hard carbon as the anodes were also evaluated. Based on the mass of cathode, the full-cells can still exhibit a high reversible capacity of ca. 131 mAh g-1. The prominent capacities make NaNi0.5Mn0.3Ti0.2O2 as an important cathode candidate for room temperature sodium-ion batteries.